The term "dioxins" is often used as an umbrella term to include different groups of structurally related chemicals, including chlorinated dibenzo para dioxins (CDDs), polychlorinated dibenzofurans (PCDFs, or “furans”), and certain dioxin-like polychlorinated biphenyls (PCBs). Each of these compounds can have varying numbers of chlorine atoms attached to the rings, which influences their toxicity.1
Chemical and Biological Properties
Most dioxins are unintentionally produced as byproducts of industrial practices, including some chemical and material manufacturing and through incineration and combustion activities.
The table below shows the chemical structure of three chemicals referred to as dioxins.2
|
TCDD ("Dioxin") |
 |
|
PCB-126 (a polychlorinated biphenyl) |
 |
|
TCDF (A furan) |
 |
2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) is the most toxic and widely studied of the dioxins. TCDD is often simply referred to as "dioxin." TCDD is a by-product formed during the manufacture of 2,4,5-trichlorophenol (2,4,5-TCP). In the past, 2,4,5-TCP was used to produce the herbicide 2,4,5-trichlorophenoxyacetic acid (2,4,5-T). 2,4,5-T was a component of Agent Orange, which was used extensively by the US military in the Vietnam War.3 It has since been banned by most countries.4
Dioxins are highly persistent and mobile. As a result, exposure can continue to be widespread long after they are produced.
Being highly lipophilic, dioxins dissolve in fat. Most dioxin exposure occurs through contaminated food. Once absorbed, dioxins are stored in animal fats. They are metabolized slowly and therefore tend to accumulate in fatty tissue and in the liver. Because of this, they bioaccumulate up the food chain. The speed of elimination of dioxins can vary with dose, quantity of body fat, age, and sex. The half-life of dioxins in the body is estimated to be 7 to 11 years.5
Health Impacts
Many dioxins and dioxin-like compounds are highly toxic to humans, although their potency varies. TCDD is the most potent and can cause adverse health effects at extremely low doses. Developing organisms, including fetuses, infants, and children, are particularly susceptible to health effects of dioxins.
Dioxins exhibit toxicity through a common mechanism of action, binding to an intracellular protein known as the aryl hydrocarbon receptor (AhR), which can alter the expression of certain genes.6
Various dioxins are carcinogens, endocrine disruptors, reproductive and developmental toxicants, and can damage the immune system.7 Data from animal models show that TCDD is linked to endometriosis and reproductive, developmental, and immunotoxic effects. Dioxins are also associated with other illnesses in people including various cancers, type 2 diabetes, ischemic heart disease, and chloracne, among others.8
Evidence links dioxins to the following diseases. Unless noted otherwise, information is from CHE's Toxicant and Disease Database. Though the database was last updated in 2016, it remains a valuable resource:9
|
Strong Evidence |
Good Evidence |
|
|
Vulnerable populations
The fetus and newborn, with rapidly developing organ systems, may be most vulnerable to dioxin exposure. Studies have shown TCDD to cause the following conditions in the offspring of exposed laboratory animals: cleft palate, skeletal deformities, kidney defects, heart malformation, abnormal prostate development, and weakened immune responses.10
Dioxin exposure, particularly through Agent Orange used in Vietnam, has been associated with birth defects in humans, including spina bifida.11
The following individuals or groups of individuals may be exposed to higher levels of dioxins, including dioxin-like PCBs:
- Those living in fenceline communities near industrial facilities, such as incinerators or chemical manufacturers.
- High consumers of fish from contaminated lakes and rivers.
- Workers in the pulp and paper industry, in incineration plants, at hazardous waste sites, and in other industries that involve processes that produce dioxins.12
- Veterans of the Vietnam War were exposed through the use of Agent Orange. More recently, over three million members of the US military have been exposed to dioxins and other pollutants from open burn pits.13
Exposures
Dioxins can be absorbed into the body by ingestion, inhalation of contaminated dust, and to some extent through the skin.14 Diet is the primary source for human exposure to dioxins, primarily through animal fats in meat and dairy products.
Human breast milk is unfortunately a contaminated food source — although, in the general population, levels have been falling in recent decades with stronger regulations on industrial dioxin emissions.15 Breastfed infants may have a daily intake of dioxins per pound of body weight 10 to 100 times greater than that of adults.16 Even though breast milk is contaminated with dioxins and other toxicants, breast milk contains many nutrients and other components highly beneficial to babies, and breastfeeding is recommended for infants for at least six months by the World Health Organization (WHO),17 the American Academy of Pediatrics,18 and the US Centers for Disease Control and Prevention.19
Dioxins have also been detected at low concentrations in cigarette smoke, home heating systems, and exhaust from vehicles. Dioxins may be formed during chlorine bleaching of pulp and paper. CDDs can be produced through burning materials that contain chlorine, such as some plastics, wood treated with pentachlorophenol (PCP), pesticide-treated wastes, other polychlorinated chemicals (such as PCBs), and even bleached paper.20
Natural occurrences such as forest fires can also produce dioxins.21 More frequent fires as a result of climate change will result in increased dioxin exposures.22
Waste incineration, including non-industrial burning, is also a major source of dioxins. Backyard burning releases pollutants at the ground level where they are readily inhaled or incorporated into the food chain.23 Increased plastic production is exacerbating this problem, particularly when the waste stream contains polyvinylchloride (PVC). As the amount of plastic in the waste stream increases, the amount of plastic in open burns of mixed waste also increases. Around the world, plastic waste is routinely burned in open fires. A recent study reported:
“The amount of plastics that get burned are estimated to be as high as the quantity of plastics emitted into the land or sea…The ash from open burning can contain dioxins, heavy metals, and other toxicants, which once settled on the ground, contaminate the soil, groundwater, and thus the organisms surrounding the environment and their respective food chains."24
Industrial incineration also continues to be a major source of dioxins, despite regulations. Technologies exist to prevent the formation and release of dioxins from industrial incineration, but emissions continue to be a problem. A 2018 report highlights the case of a “state of the art” waste incinerator built in 2011 in the Netherlands. A few years after incineration began at the facility, high levels of dioxins were detected in the eggs of chickens living nearby. Further testing verified that the dioxins in the eggs came from the incinerator.25
Some cement kilns are moving to plastic incineration as an “alternative fuel,” which some claim will reduce greenhouse gas emissions. However, EPA has found that burning plastic is not significantly better for the climate than burning coal.26 Plastics are primarily made out of fossil fuels and are known to release dioxins when burned.27
Dioxin exposures are an environmental justice issue. A 2020 report from the New School found that 79% of all municipal solid waste incinerators in the United States are located in environmental justice communities — communities of color or low-income communities that often bear a disproportionate burden of environmental harms.28
Reducing Exposures
While individuals can and should take personal steps to avoid dioxins, the possibility for consumers to reduce their own exposure is limited. This is why we need stronger environmental protections to safeguard everyone’s health.
Regulation
TCDD was not declared a probable human carcinogen by EPA until 1985. The International Agency for Research on Cancer (IARC) classified dioxin as "carcinogenic to humans" in 1997.29
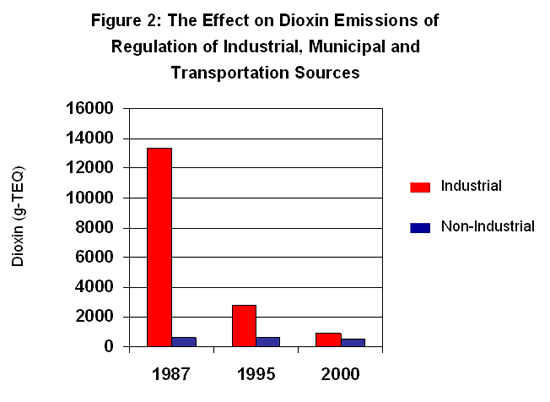
In the U.S., industrial, municipal, and transportation dioxin emissions have declined dramatically as a result of regulation, whereas emissions from backyard barrel burning of rubbish and residential wood burning have remained essentially constant since 1987.30
|
"Dioxin" here is defined as the totality of 7 dioxins and 10 furans. "TEQ" denotes "toxic equivalent," a quantitative measure of the combined toxicity of a mixture of dioxin-like chemicals. Source: Toxipedia with data from US EPA31 |
WHO's periodic studies on levels of dioxins in breastmilk provide an assessment of human exposure to dioxins from all sources. Recent exposure data indicate that measures introduced to control dioxin release in many countries have resulted in a substantial reduction in exposure over the past three decades.32
Though governments and industry have taken measures to reduce dioxin releases, their persistence in the environment and in the food chain mean that continued oversight and regulation will be needed for a long time.
In addition, increased wildfires as a result of climate change and increased plastic pollution and incineration are expected to result in more dioxins, as well as other pollutants, being unintentionally released into the environment. This is one of the multitude of reasons why tackling climate change and plastic pollution need to be a priority for governments around the world.
The Joint FAO/WHO Expert Committee on Food Additives (JECFA) performed an updated comprehensive risk assessment of PCDDs, PCDFs, and “dioxin-like” PCBs in 2001. The experts established a provisional tolerable monthly intake (PTMI) of 70 picogram/kg per month. This level is the estimate of the amount of dioxins that can be ingested over a lifetime without detectable health effects.33
EPA's National Primary Drinking Water Regulations Maximum Contaminant Level for 2,3,7,8-TCDD is 0.00000003 mg/L, or 0.003 parts per trillion.34
Personal prevention
 Reducing dioxin exposure occurs at both the individual and community level. Individuals can take these actions to reduce exposures at home:
Reducing dioxin exposure occurs at both the individual and community level. Individuals can take these actions to reduce exposures at home:
- Trim excess fat from meat
- Consume fewer high-fat dairy products
- Follow fish advisory guidelines when consuming fish and shellfish35
- Eat a balanced diet, including plenty of vegetables, plus fruits and whole grains. A diverse diet will help to avoid excessive exposure from a single source.36 .
This long-term strategy to reduce exposure is most impactful among those who may become pregnant someday, with the goal of reducing exposure of the developing fetus and when breastfeeding infants later in life.37
Community and industrial action for preventing and controlling exposure to dioxins involves measures to reduce plastic use, controls on open-pit waste burning, and prohibitions against incineration of plastic waste and other dioxin-producing materials.
This page was last revised in May 2025 by CHE’s Science Writer Matt Lilley, with input from Dr. Ted Schettler, and editing support from CHE Director Kristin Schafer.
CHE invites our partners to submit corrections and clarifications to this page. Please include links to research to support your submissions through the comment form on our Contact page.
![]()
Some information on this page is sourced from Toxipedia.