Cancer Research and Resources
Cancer is an assortment of more than 100 types of related diseases all characterized by uncontrolled cell growth. Cancerous cells have altered DNA and DNA expression. These changes can be inherited or can accrue from damage caused by environmental factors.
As cancer cells continue to grow and divide, they accrue more mutations, are less able to function as normal cells and displace healthy cells.4
![]() Benign tumors are not cancerous and may not pose a health risk, though tumors in the brain can be life threatening.5 Benign growths or tumors are usually noted by adding the ending "-oma." For example, adenoma would be a benign growth of the adrenal cortex, a hormone-producing group of cells near the kidney. Malignant tumors are noted by adding "sarcoma" or "carcinoma." A malignancy of the adrenal cortex is an adrenal cortical carcinoma. Bone cancer is osteosarcoma.
Benign tumors are not cancerous and may not pose a health risk, though tumors in the brain can be life threatening.5 Benign growths or tumors are usually noted by adding the ending "-oma." For example, adenoma would be a benign growth of the adrenal cortex, a hormone-producing group of cells near the kidney. Malignant tumors are noted by adding "sarcoma" or "carcinoma." A malignancy of the adrenal cortex is an adrenal cortical carcinoma. Bone cancer is osteosarcoma.
When a cancer spreads from its primary location, it is renamed metastatic cancer. Metastatic cancer has the same name and the same type of cancer cells as the original cancer. For example, breast cancer that spreads to and forms a metastatic tumor in the lung is metastatic breast cancer, not lung cancer.6
Incidence of Cancer
Global Incidence
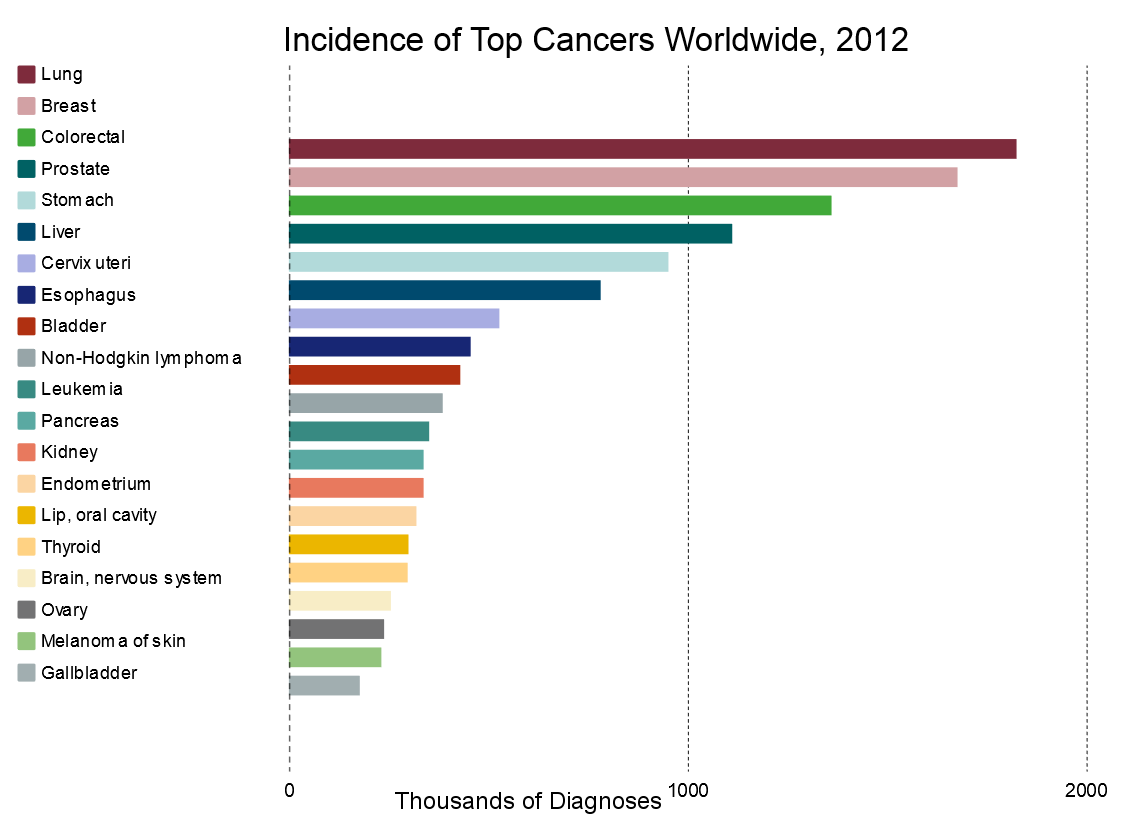
In 2012, about 14 million new cases of cancer worldwide were identified. Although total cancer incidence (number of new cases diagnosed in a year) is divided roughly equally between men and women, the types of cancer are not. Not surprisingly, men experience higher rates of prostate cancer, while women experience higher rates of breast cancer. Both experience high rates of lung and colorectal cancer.7
From Ferlay et al,8 excluding non-melanoma skin cancer; click to zoom
Denmark has the highest rate of cancer worldwide, while the US is ranked number six when both genders are combined. When genders are assessed separately, France has the highest cancer rate in men and Denmark in women.9
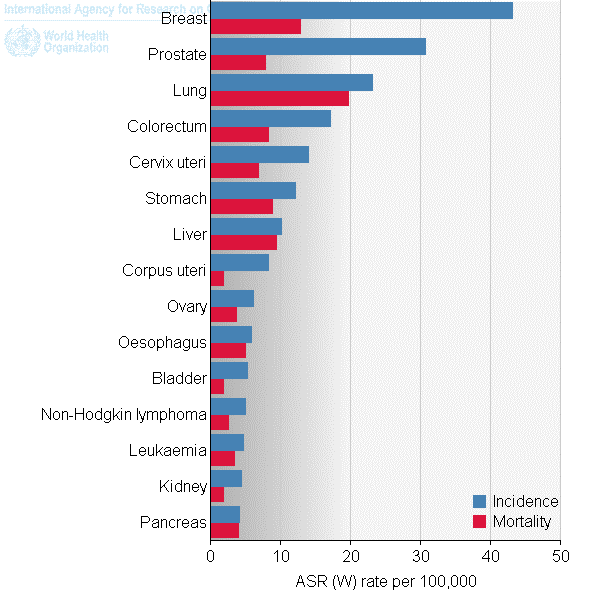
The World Health Organization provides the 2012 global age-adjusted incidence and mortality rates for the most common cancers:
| Source: International Agency for Research on Cancer (IARC).10 |
US Cancer Rates
In 2010, the President’s Cancer Panel estimated 41 percent of all Americans will be diagnosed with some form of cancer in their lifetimes. Of those, half will die from cancer. While the incidence and mortality of all cancers combined is decreasing, specific cancers, including cancers among children, are on the rise.14 Cancers increasing in the US include lung cancer (especially in women), melanoma, non-Hodgkin lymphoma and myeloma.15
Cancer Rates by Ethnicity and Gender
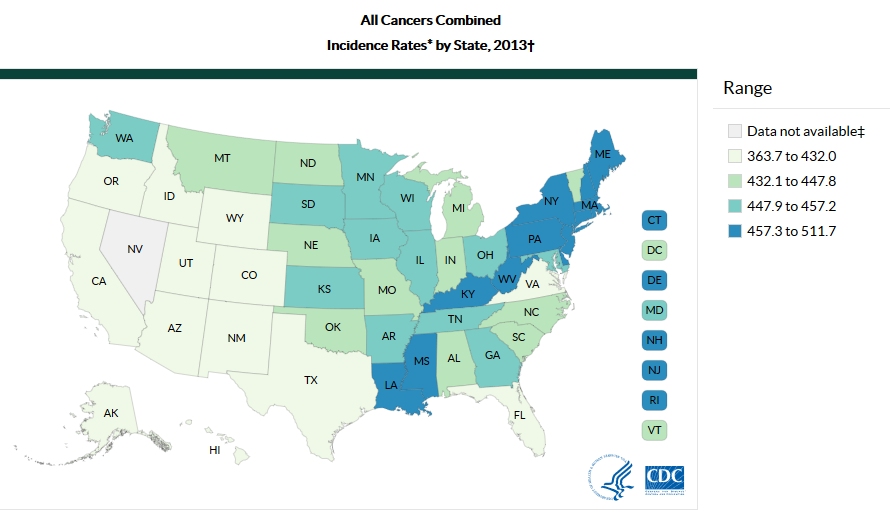
In the US, black men and white women have the highest cancer rates. The CDC provides data for US men and women by ethnicity;16 click to zoom:
Sources: CDC’s National Program of Cancer Registries and National Cancer Institute’s Surveillance, Epidemiology, and End Results program.17
*Rates are the number of cases per 100,000 persons and are age-adjusted to the 2000 U.S. standard population
†Race categories are not mutually exclusive from Hispanic origin. Rates are not presented for persons of unknown or other race. Data for specified racial or ethnic populations other than white and black should be interpreted with caution.
¶ Data are compiled from cancer registries that meet the data quality criteria for all invasive cancer sites combined for all years, 1999–2013 (covering approximately 92% of the U.S. population).
§Invasive cancer excludes basal and squamous cell carcinomas of the skin except when these occur on the skin of the genital organs, and in situ cancers except urinary bladder.
|
Stephen's story within A Story of Health highlights how environment is connected to childhood leukemia; click to open the PDF. |
Cancer in Children
Though mortality rates have decreased due to advanced therapies and early cancer detection, the incidence of cancer in children from birth to 20 years old increased between 1975 and 2006.18 Leukemias and cancers of the central nervous system (CNS, including brain cancer) were the leading diagnosed cancers in US children age 0-19 in 2013. In 2013, incidence of leukemias in children one to four years old was 8.3 per 100,000 children, while incidence of CNS cancers in children one to four years old was 4.5 per 100,000 children.19
Causes of Cancer
A wide range of chemical and physical agents, both natural and human-generated, may cause cancer. Exposure to sunlight, background radiation, natural and manufactured chemicals and even oxygen can damage our DNA and result in cancer.
![]() Various hormones are implicated in organ-specific cancer, such as breast cancer. Nutrition and diet, and specifically high caloric intake, also appear to be related to cancer, as are other lifestyle choices including tobacco use and alcohol consumption.
Various hormones are implicated in organ-specific cancer, such as breast cancer. Nutrition and diet, and specifically high caloric intake, also appear to be related to cancer, as are other lifestyle choices including tobacco use and alcohol consumption.
Certain viruses, such as the Epstein-Barr, AIDS and papilloma viruses, are known to contribute to cancer risk. Natural toxins from fungi, animals and plants may also be implicated: the grain contaminant aflatoxin B1 is known to cause liver cancer, for example.
Finally, genetics play a role, such as the known contributions of the BRCA1/BRCA2 genes in breast cancer.
Our focus is on chemical and other toxicant contributors to cancer.
Specific Toxicant Exposures Related to Cancer
There are numerous chemical exposures linked to cancer. CHE’s Toxicant and Disease Database lists hundreds of specific disease-toxicant associations. We have highlighted just a few exposures linked to common cancers:
|
Some Examples of Toxicant Connections to Common Cancers |
|
|
Type of Cancer |
Toxicant |
|
Strong Evidence |
|
|
Breast cancer |
Estrogens Tobacco, active smoking and secondhand |
|
Lung cancer |
Aluminum Cadmium Chromium (IV) Mineral oils Tobacco, active smoking and secondhand |
| Good Evidence | |
|
Breast cancer |
PCBs Solvents |
| Childhood brain cancer | |
| Leukemias | Pesticides |
|
Lung cancer |
Copper Solvents |
|
Prostate cancer |
Pesticides |
Toxicant Classification
|
Air Pollution
Exhaust from cars, trucks, ships and other vehicles, especially diesel particulates, cause up to 30 percent of air pollution-related cancers. Diesel particulate is a Group 1 carcinogen22 with the average lifetime risk for cancer attributable as high as 1 in 1,000 people.23
Industrial combustion also creates particulate matter associated with lung cancer. This pollution can travel through the air to nearby schools, exposing children. A 2008 investigation of almost 128,000 schools found that the majority experience toxic air pollution, and one in six schools were located within a mile of a major industrial plant.24
Asbestos
Asbestos, a natural mineral fiber used in many industrial materials and products, continues to cause serious human health effects. Inhalation of asbestos is the primary cause of mesothelioma, cancer of the membrane that covers and protects most of the body’s internal organs. IARC labels asbestos as a Group 1 carcinogen,25 and some evidence indicates it increases the risk of other cancers besides mesothelioma. More than 70 percent of patients with mesothelioma cancer have a history of exposure to asbestos at work.26
Benzene
![]() Benzene is widely used in the production of other products such as rubber, nylon, synthetic fiber, lubricants, glues, detergents, dyes, drugs and pesticides; it is also part of vehicle exhaust and cigarette smoke.
Benzene is widely used in the production of other products such as rubber, nylon, synthetic fiber, lubricants, glues, detergents, dyes, drugs and pesticides; it is also part of vehicle exhaust and cigarette smoke.
Benzene is classified as a Group 1 human carcinogen by IARC.27 Liver enzymes convert benzene to more toxic metabolites, which is thought to be what causes its carcinogenicity. Chronic exposure to benzene affects the bone marrow by crippling blood cell production, causing anemia, which can ultimately result in leukemia.
Formaldehyde
A toxicant produced by the manufacturing industry, formaldehyde can be found in building materials. IARC classifies formaldehyde as a Group 1 human carcinogen for nasal and nasopharynx cancer. IARC also concluded that there is strong but not sufficient evidence for a causal association between leukemia and occupational formaldehyde exposure.28
Metals
Arsenic is one of the most serious human carcinogens. Inorganic arsenic can be found naturally in drinking water and is associated with skin, lung, bladder and kidney cancer in both men and women and prostate cancer in men.29 IARC classifies arsenic as a Group 1 carcinogen.30
Hexavalent chromium, another IARC Group 1 carcinogen,31 damages DNA and can cause nasal and lung cancer. Exposure is known to occur in occupational settings (such as manufacturing, steel and alloy production and leather tanning) and through communitywide exposure from improper disposal practices.32
Cancers Associated with Pesticides33
|
Pesticides
There are hundreds of pesticides in many classes, each with different toxicological effects. Thus is it an oversimplification to say that pesticides as a whole cause cancer. However, exposures to certain pesticides are associated with several types of cancers (see box to the right). IARC has classified many pesticides as known, probable or possible human carcinogens (Groups 1, 2A and 2B).34 Some examples of these pesticides are listed in the box above detailing IARC's classifications.
Pharmaceuticals
IARC classifies these pharmaceutical drugs or treatments as Group 1 carcinogens:35
Treatment as Risk FactorMany cancer treatments have serious toxicological side effects. IARC classifies MOPP and other combined chemotherapy including alkylating agents as a Group 1 carcinogen. Radiation therapy employs ionizing radiation, also a Group 1 carcinogen.36 Risks from chemotherapy can extend beyond cancer patients to family members, nurses and others who are exposed to the chemicals or to body fluids containing residues of drugs.
|
- Azathioprine
- Busulfan
- Chlorambucil
- Chlornaphazine
- Cyclophosphamide
- Cyclosporine (cyclosporin)
- Diethylstilbestrol
- Estrogen-progestogen oral contraceptives (combined)
- Estrogen-progestogen menopausal therapy (combined)
- Estrogen therapy, postmenopausal
- Etoposide (also in combination with cisplatin and bleomycin)
- Melphalan
- Methoxsalen (8-methoxypsoralen) plus ultraviolet A radiation
- MOPP and other combined chemotherapy including alkylating agents
- Phenacetin
- Semustine (methyl-CCNU)
- Tamoxifen
- Thiotepa
- Treosulfan
Polyhalogenated Biphenyls
Polychlorinated biphenyls (PCBs) and polybrominated biphenyls (PBBs) were used in electrical equipment, manufacturing and industrial processes, although PCBs were banned from manufacture n 1977.37 High rates of PBB in the blood are associated with several cancers including breast, lymphoma (non-Hodgkin) and several digestive cancers. PCBs are associated with liver cancer and are a suspected contributor to breast, skin and blood cancers. Some research demonstrates that children exposed to PCBs in the home are more likely to develop leukemia than children who are not.38
Radiation
Ultraviolet radiation, UVA and UVB, is emitted by tanning beds, sun lamps and the sun itself. Extensive evidence has demonstrated that all can cause skin cancer.39
|
image from Jlhopgood at Creative Commons |
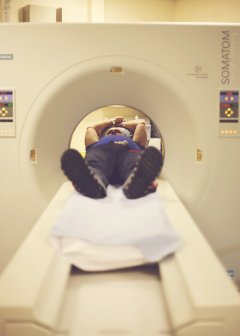
Medical Radiation. By 2006, almost half of the typical American exposure to ionizing radiation had come from medical devices, especially from computed tomography (CT) scans. These exposures are associated with an increase in cancer risk. An adult receiving an annual CT scan for a 30-year period is projected to increase his or her overall lifetime risk of cancer mortality by almost two percent. Though it might not be significant at the individual level, it is a considerable concern across the population of patients receiving CT scans on a regular basis. Scans of young children, who are more vulnerable to radiation's effects than adults, are even more concerning.40
Substantial evidence demonstrates that medical radiation is an important and controllable cause of breast cancer.41
Radon is another source of radiation known to cause cancer (IARC Group 1 carcinogen),42 with between 15,000 and 22,000 lung cancer deaths in the United States each year related to radon.43 For information on non-ionizing radiation, please see Electromagnetic Energy below.
Solvents
Perchloroethylene (PCE) used by 28,000 dry cleaners in the US is labeled as “reasonably anticipated to be carcinogenic to humans” by the US National Toxicology Program (NTP) and is an IARC Group 2A carcinogen.44 The dry cleaning process exposes workers and releases significant amounts of PCE into the air, and improper storage and disposal of PCE has contaminated soil and water across the US. Breast cancer is specifically associated with PCE in drinking water.45
Trichloroethylene (TCE), used in manufacturing, is labeled reasonably anticipated to be a human carcinogen by the (NTP)46 and is an IARC Group 1 carcinogen.47 TCE is strongly associated with kidney and liver cancer and is suspected to be associated with lymphomas, leukemia and cervical cancer. Improper disposal has released TCE into water sources, and it has been detected in up to 34 percent of US drinking water.48
Other Contributors to Cancer
Hormones
![]() Some cancers, including breast, endometrium, ovary, prostate, testis and thyroid, are hormone-related. Naturally occurring hormones, hormone treatments, or environmental chemicals that mimic or otherwise alter hormone functions (EDCs) can play a role in cancer development and increase risk.
Some cancers, including breast, endometrium, ovary, prostate, testis and thyroid, are hormone-related. Naturally occurring hormones, hormone treatments, or environmental chemicals that mimic or otherwise alter hormone functions (EDCs) can play a role in cancer development and increase risk.
Lifestyle Choices
|
image from Andréia Bohner at Creative Commons |
Lifestyle choices have been a main driver of many cancers. Tobacco use kills around six million people worldwide each year, with more than five million of those deaths the result of direct tobacco use. More than 600,000 deaths are the result of exposure to secondhand smoke among non-smokers.49
Consumption of alcoholic beverages causes cancer of the mouth, pharynx, larynx and esophagus50 and contributes to the risk of liver cancer.51
|
image from Michael Johnson at Creative Commons |
Both overweight/obesity52 and consumption of specific foods are associated with some cancers. Processed meats (Group 1) and red meats (Group 2A) are classified as carcinogens by IARC.53 Many other foods and micronutrients (such as vitamin D or calcium) are thought to either increase or reduce cancer risk, with supporting evidence in animal studies, but research is limited or inconclusive about effects in humans.54
Poverty and Neighborhoods
While some have concluded that poverty is a risk factor for cancer,55 a 2014 analysis in the United States found that income level has a complex relationship with cancer incidence. For all cancer types combined in that investigation, cancer incidence was negligibly associated with poverty at a neighborhood level (not individual level); however, 32 of 39 cancer types showed a significant association with poverty (14 positively associated and 18 negatively associated). Certain cancers—Kaposi sarcoma and cancers of the larynx, cervix, penis and liver—were more likely in the poorest neighborhoods, while other cancers—melanoma, thyroid, other non-epithelial skin and testis—were more likely in the wealthiest neighborhoods.56
A study published in 2011 reported that living in more highly segregated areas with higher crime rates is linked to an increased risk of developing cancers of all kinds in adults 55 and older, for Whites as well as Blacks, and for both men and women. Income was not the driver, for the researchers stated that "segregation and crime increased the chances of developing cancer even after we controlled for socioeconomic resources at both the individual and the neighborhood level."57 A 2015 review described analyzed results of 34 studies (including the 2011 study above) and found "evidence that social and built environment attributes exert independent influences beyond SES [socioeconomic status] on cancer incidence, tumor characteristics, treatment, survivorship and survival/mortality."58
Neighborhoods that are adjacent to or downwind of refineries, coal-fired power plants and other industries that emit large quantities of carcinogens often experience elevated cancer rates. These are referred to as "fenceline communities" and are considered as cumulative impacts.
Infectious Disease
Some infectious diseases, both viral and bacterial, are associated with cancer:59
- Certain strains of human papilloma viruses (HPVs) are linked to cancers of the cervix, vagina, vulva, penis, anus, and some cancers of the mouth, throat, head and neck.
- Epstein-Barr virus (EBV) is linked to nose and throat (nasopharyngeal) cancer, lymphoma of the stomach, Hodgkin lymphoma and Burkitt lymphoma.
- Hepatitis B virus (HBV) and hepatitis C virus (HCV) are linked to chronic liver infections, which can raise the risk of liver cancer (hepatocellular carcinoma).
- Human herpes virus Type 8 (HHV-8), also called Kaposi sarcoma herpes virus (or KSHV), is linked with a type of cancer called Kaposi sarcoma. Most people with HHV-8 do not develop Kaposi sarcoma unless they are also infected with human immunodeficiency virus (HIV), the virus that causes AIDS.
- Human T-lymphotropic virus-1 (HTLV-1) is linked with certain types of lymphocytic leukemia and non-Hodgkin lymphoma (NHL).
- Helicobacter pylori is linked to gastric cancer.60
Emerging Concerns
Electromagnetic Energy/Non-Ionizing Radiation
Electromagnetic (EM) Energy, specifically non-ionizing radiation, comes from wired and wireless devices. Cell phones and their towers emit radiofrequency radiation (RF) while power lines and appliances emit extremely low-frequency electromagnetic fields (ELF-EMF). The ubiquitous use of cell phones raises concern over the cancer risk posed by exposure to the EM energy emitted by these devices, and a growing body of evidence suggests that EMR/EMF may have deleterious effects on human health with prolonged exposure.61 IARC has classified radiofrequency electromagnetic fields as possibly carcinogenic to humans (Group 2B).62
Nanotechnology
Engineered nanomaterials (ENM) can be found in many consumer products including sunscreens, cosmetics and other personal care products, clothing and food storage containers. Research about their safety has lagged behind the production of these particles, but their small size and ability to penetrate cells in the body makes this technology an area of concern.63
Combined and Interacting Effects
|
image from Rafael Mikaelyan at Creative Commons |
Chemical mixtures or exposure to multiple agents can increase the incidence of cancer. For example, exposure to cigarette smoke together with exposure to asbestos leads to a greatly increased risk of lung cancer.64 The risk of lung cancer from radon exposure is estimated at between 10 to 20 times greater for persons who smoke cigarettes as compared with those who have never smoked.65 The risk of cancer from consumption of alcoholic beverages is most pronounced among smokers.66 Some evidence suggests that diets deficient in micronutrients such as vitamins B2, B6, B12 and folic acid may increase susceptibility to arsenic-induced cancers.67
President’s Cancer Panel
|
In 1971, President Richard Nixon signed the National Cancer Act into law, establishing the President’s Cancer Panel. This panel is tasked with gathering input from stakeholders to form recommendations for the US President to consider. The panel publishes a report every two years. In 2010, the panel's report, titled Reducing Environmental Cancer Risk: What We Can Do Now, was informed by 45 experts spanning government, industry, academia and advocacy communities. This summary webpage draws heavily from that report, with updates as available. |
The panel's 2010 report indicated specific barriers to further understanding and acting on the relationship between environmental exposures and cancer:68
- Low priority and lack of research on the environmental causes of cancer
- The limited number of environmental oncologists
- Variation in efforts to identify and quantify environmental exposures
- Failure to account for low-dose exposures
- Failure to review in utero, childhood and lifelong exposures
- The reactionary approach of US chemical policy
- The fragmented nature of regulatory agencies
The panel's conclusions:
- The nation needs a comprehensive policy agenda for environmental contaminants.
- Children are at special risk and should be protected.
- Continued health research in humans is needed.
- A new paradigm is needed for long-latency diseases.
- Existing regulation must be enforced and updated.
- Radiation exposure from medical sources is underappreciated.
- The medical community should consider occupational and environmental sources in diagnosis.
- Full disclosures to workers and the general public should be made regarding environmental cancer risks.
- The military should aggressively address the toxic environmental exposures it has caused.
- Safe alternatives to current chemicals are urgently needed.
Vulnerable Populations
|
A patient at the Children's Cancer Center in Bishkek is held in her mother's arms; photo from DVIDSHUB at Creative Commons. |
Children are exposed to carcinogenic chemicals through every route of exposure (air, water, food) and in the many environments they experience daily (school, home, cars). They are at increased risk for several reasons: Because children are smaller and known to intake more air and water for body size versus adults, they receive a higher dose of exposure than their adult counterparts. The metabolic pathways (routes of detoxification in the body) are not fully developed in the first few years of life, potentially amplifying any negative effects of exposure. Finally, cancerous changes that start in childhood have an entire lifetime to manifest and propagate in the body. Thus the younger a person is when exposed, the more opportunity there is throughout the lifespan for cancer to develop. This is especially concerning given the increasing use of CT scans and nuclear medicine for diagnosis and treatment of childhood illness.69
Occupations with higher exposures to environmental cancer-causing agents include mining, manufacturing, construction, agriculture and certain service industries. These jobs are often held by people from disadvantaged populations, making environmentally caused cancer outcomes an issue of health equity and environmental justice.70
IARC list these occupations as having increased risk of cancer, usually due to exposures to chemicals:71
| Occupation | Classification |
| Applicator of non-arsenical insecticides | 2A |
| Chimney sweep | 1 |
| Dry cleaner | 2B |
| Firefighter | 2B |
| Graphite and silicon carbide synthesis work with Acheson process | 1 |
| Hairdresser or barber | 2A |
| Iron and steel founding work | 1 |
| Painter | 1 |
| Petroleum refining work | 2A |
| Printing processes work | 2B |
| Road paver with straight-run bitumens | 2B |
| Roofer with oxidized bitumens | 2A |
| Shiftwork that involves circadian disruption | 2A |
Ashkenazi Jews—members of the Jewish community who trace their roots to Central or Eastern Europe—have much higher rates of specific mutations in BRCA1 and BRCA2 genes than the general population. These mutations increase the risk of some cancers, including breast and ovarian in women and breast and prostate in men.72
Women carry a larger body burden of endocrine disrupting chemicals, placing them and their developing children at a higher risk of cancers related to hormone disruption.73
People who have received an organ transplant are at higher risk of some cancers, thought to be due to immunosuppression, impaired DNA damage repair and infection with oncogenic viruses.74
Genetic Toxicology and the Biology of Cancer
![]() Genetic makeup can increase the likelihood that certain cancers will develop. For example, about 12 percent of American women will develop breast cancer sometime during their lives, but the most recent estimates are that 55 to 65 percent of women who inherit a harmful BRCA1 mutation and around 45 percent of women who inherit a harmful BRCA2 mutation will develop breast cancer by age 70.75
Genetic makeup can increase the likelihood that certain cancers will develop. For example, about 12 percent of American women will develop breast cancer sometime during their lives, but the most recent estimates are that 55 to 65 percent of women who inherit a harmful BRCA1 mutation and around 45 percent of women who inherit a harmful BRCA2 mutation will develop breast cancer by age 70.75
DNA damage occurs regularly as part of the cell process and from interaction with both normal cellular chemicals and with toxic chemicals. A robust repair mechanism rapidly repairs DNA damage most of the time. However, if the DNA is repaired incorrectly, a mutation occurs. Many mutations have no effect, some have minor effects, and a small number have life-threatening effects. If a mutation occurs in our germ line cells, the mutation can be passed on to our offspring. Some mutations cause a cell to divide uncontrollably, becoming a malignant cell and causing cancer.
Mutagens
Ames AssayIn 1946 nitrogen mustards (derived from mustard gas first used by the military in 1917 during WWI) were shown to induce mutations in fruit flies and reduce tumor growth in mice. Genetic toxicology developed ways to test chemical and physical agents for their mutagenic properties, and in the 1970s Bruce Ames and others developed a cellular-based test for genetic mutations. This test became known as the Ames assay. Sophisticated variations of these tests are now required by many government regulatory agencies to test chemicals for mutagenicity before they are approved for use. |
![]()
Chemicals that induce mutations in DNA are called mutagens, and when these changes lead to cancer, the chemical is called a carcinogen. Not all mutagens are carcinogens, and not all carcinogens are mutagens.
Often a metabolite of a compound causes cancer, not the original compound. Ideally, a foreign chemical is made less toxic when metabolized, but sometimes a chemical can be made more toxic. This more-toxic chemical can then interact with cellular DNA or proteins and produce malignant cells. This process is called bioactivation. A chemical can encourage bioactivation or accelerate the development of a cancer. Many variations of the Ames assay that include liver cells were developed to simulate the metabolism of the chemical in the liver and determine if bioactivation would result in mutations.
Efforts to understand the underlying biology of cancer are ongoing. Genomic science is helping to explain why some people are more susceptible to cancer than others. However, cancers resulting from single genes inherited from parents comprise fewer than five percent of all cancers in the US.76 Environmental insult is clearly involved in many—perhaps most—cancers.
The genetic changes that contribute to cancer tend to affect three main types of genes—proto-oncogenes, tumor suppressor genes and DNA repair genes.81
- Proto-oncogenes are involved in normal cell growth and able to morph into cancer-causing genes (oncogenes).
- Tumor suppressor genes control normal cell growth and division.
- DNA repair genes are involved in fixing mutations that can lead to cancer.
Epigenetics
Healthy gene expression is critical to normal cellular processes including cell growth and normal cell death. Environmental contaminants can interfere with normal biological processes by changing the chemical tags on DNA that regulate gene expression. This is known as epigenetic modification. Through changes in the epigenetic marks on cancer-related or cellular cycle genes, genes that suppress tumor growth or programmed cell death may be silenced, while genes that promote cellular growth may be overexpressed.
Environmental toxicants can also impact appropriate cellular migration during development, as was the case with diethylstilbestrol (DES). This drug was prescribed to thousands of pregnant women between 1938 and 1971 for the prevention of miscarriages. We now know that an epigenetic effect of this drug inhibits the proper development of the reproductive tract in the developing fetus, especially in females, and increases the risk of rare types of vaginal and cervical cancer in daughters. There is some data to suggest this epigenetic effect could also be inherited by the granddaughters of the original DES patients.82
Prevention
Steps can be taken individually to reduce exposures to carcinogenic chemicals. Because children are more vulnerable to these exposures, child care providers should look to reduce or remove exposures to toxics. Here are some recommendations from the President’s Cancer Panel:83
- Remove shoes before entering the home.
- Wash contaminated clothing separately from other family laundry.
- Filter drinking water (preferable to commercially bottled water).
- Store and carry water in stainless steel, glass, or phthalate-free containers.
- Microwave foods in ceramic or glass.
- Wash conventionally grown food before eating and purchase organic when possible and appropriate.
- Eat free-range meat low in antibiotics and growth hormones if possible.
- Avoid eating processed, charred and well-done meats.
- Consult the Consumer Products Information Database (CPID) to make informed decisions when purchasing products.
- Properly dispose of pharmaceuticals, household chemicals, paints and other soil contaminants.
- Turn off lights and electrical devices when not in use.
- Use buses, bicycles, fuel efficient cars or walk when traveling.
- Avoid smoking and exposure to secondhand smoke.
- Reduce electromagnetic radiation exposure by wearing a wired cell phone headset and keeping calls short.
- Check home radon levels and reduce levels if needed.
- Be aware of radiation exposure from medical sources.
- Wear protective clothing and sunscreen while outdoors and avoid tanning beds.
History
![]() The oldest descriptions of cancer dates back to Egypt, around 1600 BC. The so-called Edwin Smith Surgical Papyrus describes eight cases of what appears to be breast cancer. The tumors of the breast were treated by cauterization, with a tool called "the fire drill."
The oldest descriptions of cancer dates back to Egypt, around 1600 BC. The so-called Edwin Smith Surgical Papyrus describes eight cases of what appears to be breast cancer. The tumors of the breast were treated by cauterization, with a tool called "the fire drill."
|
Chimney sweep in 1850 |
The first recorded occupational cancer exposures occurred in 1775, when the English physician and surgeon Percivall Pott made the observation that exposure to soot might explain the high incidence of scrotum cancer in chimney sweeps. This was the first indication that exposure to chemicals, in this case a complex mixture, could cause cancer. However, this new knowledge did not immediately translate into improved working conditions for chimney sweeps. More than 100 years later, cancer of the scrotum was rare in continental Europe but still high in England, possibly due to better hygiene practices in Europe.
Early epidemiological studies indicated that inorganic metals such as arsenic and nickel could cause cancer; this was subsequently confirmed in animal studies.
The industrial revolution of the late 19th and early 20th centuries brought clear confirmation that occupational exposure to chemicals could cause cancer. The first indication came from increases in skin and bladder cancers associated with cutting oils and dyes. In 1895, bladder cancer was associated with workers in the aniline dye industry. Further worker-based studies found that exposure to specific chemicals could be responsible for the cancer. In 1915, Japanese researchers reported that they could induce skin tumors in animals by repeatedly applying a coal tar solution to the skin of rabbits. These early studies, subsequently repeated with mice, ushered in the scientific investigation of the chemical causes of cancer. These studies also initiated the systematic investigation of the adverse health effects of chemicals, which in many ways laid the foundation for the toxicological sciences.
|
Marie Curie, image from Tekniska museet at Creative Commons |
Chemicals are not the only cause of cancer. Marie Curie, awarded Nobel Prizes in both physics and chemistry, discovered radium in 1898. The green glow of radium fascinated people, and it was touted as a cure for many diseases, including cancer, rheumatism, syphilis, multiple sclerosis, sexual dysfunction, hypertension, arthritis, neuritis, diabetes and anemia.84 Now radium-224, radium-226, radium-228 and their decay products are all listed by IARC as Group 1 carcinogens.85
Lung diseases possibly related to radon were first reported in the 1400s, and in 1879 lung cancer was seen in European miners. Radon was discovered several years later in 1900 by the German chemist Friedrich Ernst Dorn. Regulation of workplace exposure began in the 1950s and subsequent studies of underground mine workers in Canada, the Czech Republic, France, Australia, Sweden and the United States have allowed researchers to develop very sophisticated models of the cancer-causing effects of radon.
The use of nuclear weapons by the US military in World War II, and subsequent development of the defense and nuclear industries in various countries, have raised public awareness of the health consequences of radiation exposure. Naturally occurring background radiation, as well as our many medical and industrial exposures to radiation, are responsible for some cancers.
Research continues to expand and refine our understanding of cancer's causes.
|
History of Carcinogen Identification |
||
|
Year |
Cancer Type |
Cause |
|
1775 |
Scrotal cancer |
Soot |
|
1822 |
Skin cancer |
Arsenic |
|
1879 |
Lung cancer |
Uranium mining |
|
1895 |
Bladder cancer |
Aniline dyes |
|
1902 |
Skin cancer |
X-rays |
|
1908 |
Leukemia |
Filterable agents |
|
1914 |
Skin cancer (experimental induction) |
Coal tar |
|
1928 |
Skin cancer (experimental induction) |
UV light |
Ethics of Gene Testing
The BRCA1 and BRCA2 genes, first discovered in the mid 1990s, are the most studied genes related to breast and ovarian cancer. Both genes came to public attention in 2013 when Angelina Jolie, a popular US actor, announced she tested positive for a faulty BRCA1 gene, which increased her risk for breast and ovarian cancer.86 This story has motivated other women to get genetic testing, but the results of these tests are not always clear.
With the rise of multi-gene panels and whole genome sequencing, lab tests uncover variants in these two genes that have not been categorized as causing disease, known as variants of uncertain significance (VUS). A provider receiving a VUS result for a patient does not have a clear clinical course of action to recommend and thus might recommend unnecessary preventive surgeries. Patients receiving VUS receive no additional information on their genetic risk status, which can cause an increase in anxiety or worse as they misunderstand the meaning of the test result.87
As research advances, previous VUS results might be updated to be deleterious. It is unclear who is responsible for reviewing these updates, how providers might keep up with these updates and how patients might receive updates of their test results.88
For decades the patent on BRCA1/2 testing was held by Myriad Genetics, who charged significant fees for these tests. Since they held the patent, they also held the largest database of results from which VUS could be studied. This slowed the progress of VUS classification for the broader population. When in 2013 the US Supreme Court ruled that genomic DNA could not be patented, BRCA1/2 testing and research on results could be done by other certified laboratories. This enabled other entities to develop databases where VUS could be studied and classified.89
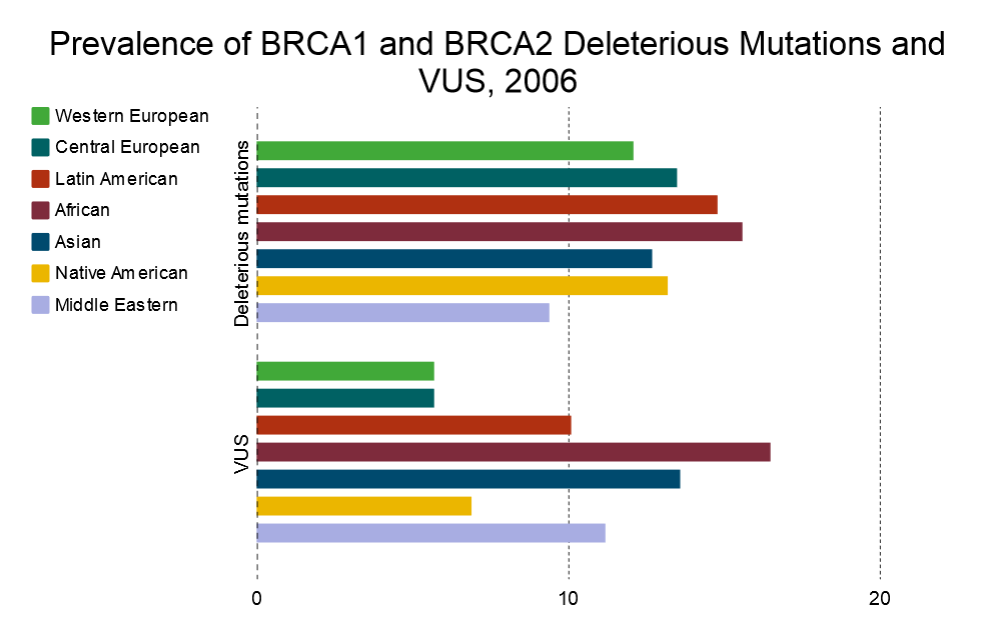
VUS occur more in non-Caucasian ethnicities. A 2009 study found that, compared to western or central European populations, the VUS rate in Latin American, African, Asian and Middle Eastern populations was from 77 to 189 percent higher:90
Persons with incomplete personal or family cancer history were excluded. Ashkenazi Jews were excluded from this analysis because testing procedures are substantially different in this group; click to zoom.
There are a few aspects of genetic testing that cause concern:
- First, 85 percent or more of breast cancer cases are estimated to result from non-genetic factors.91 Patients might misinterpret negative genetic test results to mean they are not at risk of breast cancer when that is clearly not the case.
- Second, family history can be more informative than genetic testing regarding a patient’s risk for genetically derived breast cancer.
- Third, VUS can lead to misunderstanding among patients and the medical community, and there is no clarity regarding the obligation to follow up when new information is available. There is not even a standard classification system for test results.92
- Finally, the increased rate of VUS results in non-white populations could be because these populations are underrepresented in reference databases. The inequity of research in these populations could result in a disproportionately high VUS rate.93
See more about environmental influences on cancer in the list of CHE publications and Dig Deeper resources in the right sidebar.
CHE’s Work on Cancer
Consensus Statements
CHE Consensus Statement on Cancer and the Environment, 2008. In partnership with the Lowell Center for Sustainable Production and the Breast Cancer Fund, CHE developed a consensus statement that lays out the scientific rationale for stronger cancer prevention and enumerates specific research and policy initiatives to prevent environmental exposures that contribute to cancer.
Consensus Statement on Breast Cancer and the Environment, 2006. CHE’s Breast Cancer and the Environment Working Group (now merged into the Cancer ScienceServ) created this statement calling for testing of chemicals for their effects on health and the environment before they are marketed and also advocating that patient and health professional organizations add primary prevention to organizational agendas.
President’s Cancer Panel
 A past major activity of the CHE Cancer Working Group was its involvement in the President's Cancer Panel. Starting in September 2008, the panel convened public meetings focusing on cancer and the environment. CHE Partners—including Jeanne Rizzo, Dr. Dick Clapp, Dr. Devra Davis, Dr. Phil Landrigan, Dr. Sandra Steingraber, and Dr. Tyrone Hayes—presented evidence to both the panel and the American people on what was known about a variety of environmental contributors of cancer.
A past major activity of the CHE Cancer Working Group was its involvement in the President's Cancer Panel. Starting in September 2008, the panel convened public meetings focusing on cancer and the environment. CHE Partners—including Jeanne Rizzo, Dr. Dick Clapp, Dr. Devra Davis, Dr. Phil Landrigan, Dr. Sandra Steingraber, and Dr. Tyrone Hayes—presented evidence to both the panel and the American people on what was known about a variety of environmental contributors of cancer.
The full 2010 report, Reducing Environmental Cancer Risk: What We Can Do Now, is available on the President's Cancer Panel page on the National Cancer Institute website. The report, which informs the National Cancer Program, has brought unprecedented attention to the environmental exposures that increase cancer risk.
Margaret Kripke, PhD, co-author of the 2010 President's Cancer Panel report, was interviewed by The New School at Commonweal on February 7, 2011. Listen to the audio recording.
This page's content was created by Lorelei Walker, PhD, and Nancy Hepp and last revised in September 2016. Content from Toxipedia is noted with the logo.
CHE invites our partners to submit corrections and clarifications to this page. Please include links to research to support your submissions through the comment form on our Contact page.
* header image from Jess Sloss at Creative Commons